Androgenetic alopecia (AGA) affects millions of men and women worldwide, causing progressive hair loss that goes beyond aesthetics: it impacts self-esteem, confidence, and quality of life. As one of the most common causes of hair loss, AGA represents a key segment of the global hair health market, where innovation can make a real difference.

Until now, available treatments have focused on:
Such as minoxidil or finasteride, which offer variable results and may cause side effects.
Such as hair transplants, which are invasive, require long recovery times, and do not always guarantee natural-looking results.
Such as PRP, with limited effectiveness and unpredictable outcomes.
While these options have helped many patients, they do not address the follicle’s microenvironment in a comprehensive way, limiting their effectiveness and acceptance. These limitations highlight the need for innovative, safe solutions that better meet patients’ real needs.
In this context, AGAcell® emerges as the first preclinical research program exploring an allogeneic mesenchymal stem cell-based therapy for androgenetic alopecia.
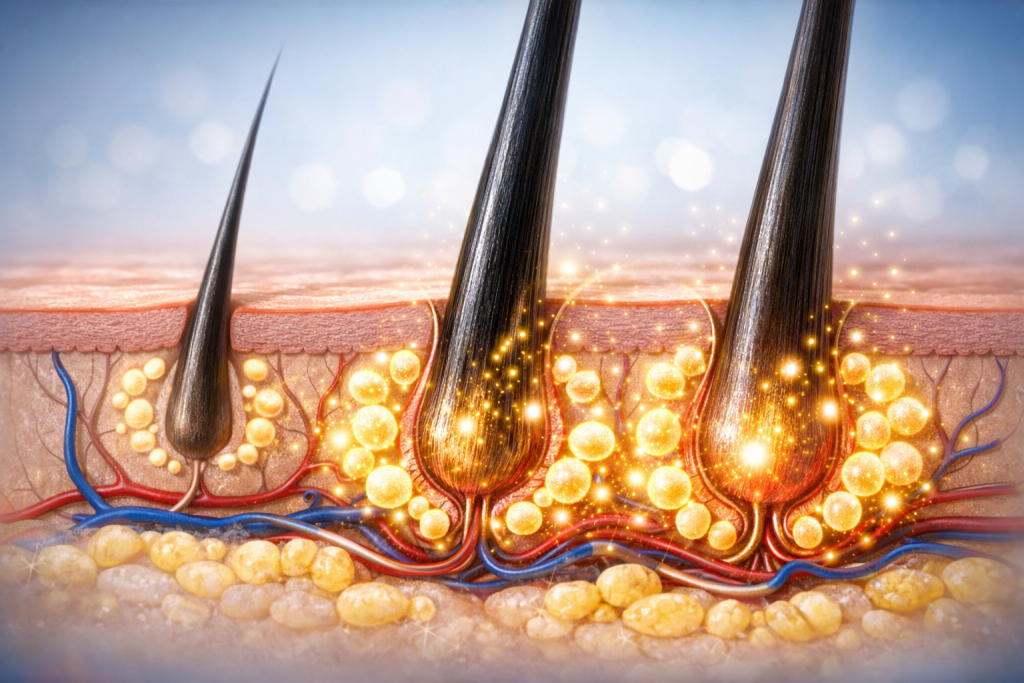
AGAcell® is designed to restore the follicle microenvironment and reactivate its key functions, promoting long-term hair growth through a minimally invasive, non-surgical procedure.
The AGAcell® formulation combines:
GMP grade, capable of regenerating and protecting the follicle.
A molecule that enhances cellular energy and supports the regeneration of the follicle microenvironment.
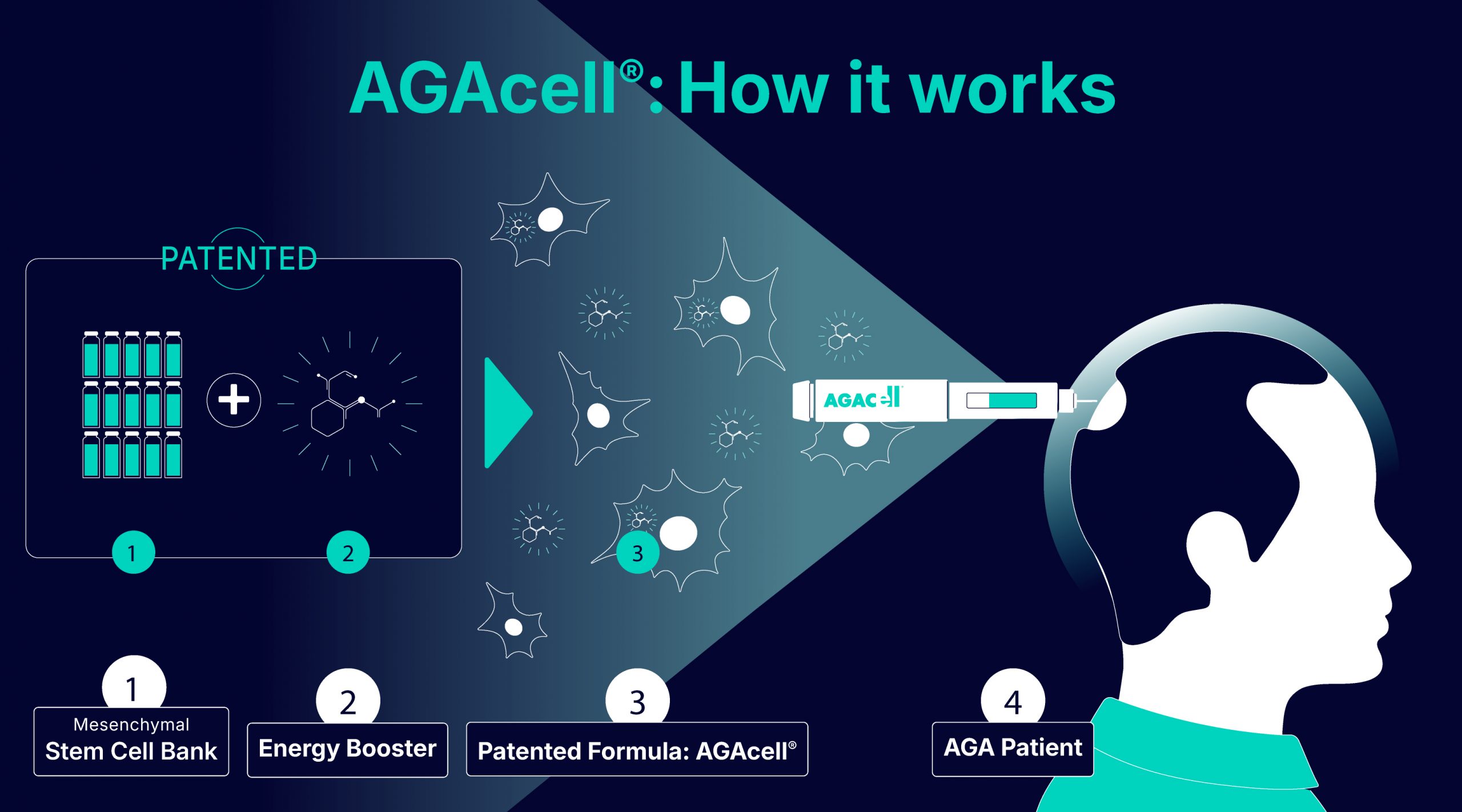
Our GMP compliant manufacturing process starts with the screening of a healthy donor, from whom we extract and isolate mesenchymal stem cells. Subsequently, we perform cell expansion under rigorously controlled conditions to achieve an optimal quantity of viable cells which are then cryopreserve in a mesenchymal stem cell bank.
Mesenchymal stem cells (MSCs) are being investigated for their capacity to interact with the resident follicular cells and support regeneration of the hair-follicle niche. AGAcell® combines these allogeneic MSCs with a bioactive molecule that synergistically enhances their regenerative activity.
The clinical planned procedure involves intradermal administration into the affected scalp area by a qualified dermatologist. Ongoing studies aim to determine whether this minimally invasive approach can modulate the follicle’s cellular niche, restoring hair-growth potential and long-lasting solution for AGA.
Androgenetic alopecia is not just an aesthetic issue. Its progression causes:
Loss of the scalp’s natural UV protection, increasing the risk of photoaging and melanoma.
Progressive follicle miniaturization, driven by dihydrotestosterone (DHT) and the activation of androgen receptors in dermal papilla cells.
Deterioration of the follicle microenvironment, due to oxidative stress, inflammation, and reduced vascularization.
AGAcell® offers a comprehensive and regenerative approach, addressing both hair loss and the overall health of the follicle and scalp. Thanks to its innovative approach, androgenetic alopecia is no longer just an aesthetic problem—it becomes a target for advanced hair restoration and regeneration
As a potential allogeneic therapy, AGAcell® is based on mesenchymal stem cells sourced from a qualified donor and stored in a cell bank. Unlike autologous procedures, where cells are harvested from, and later re-administered to, the same patient, an allogeneic strategy may offer several practical advantages:
Allogeneic MSCs are harvested from rigorously screened donor tissue and expanded under standardized GMP conditions, yielding more consistent viability and potency than autologous cells, whose quality can vary with each patient’s age, comorbidities and disease state.
Cells from a single donor batch can be expanded under GMP conditions for multiple doses, which could reduce per-patient manufacturing costs compared with one-patient-one-batch autologous processes.
The envisaged protocol requires only a single visit for intradermal administration; autologous approaches usually need a first visit for cell harvest and a second for reinfusion. Recovery is much faster and less painful compared to surgical hair transplants.
As a cryopreserved, “ready-to-use” product, treatment could in principle be scheduled soon after prescription, without the lead time needed to culture each patient’s own cells.
AGAcell® is planned to be manufactured under GMP compliant conditions, meeting the same quality and safety requirements applied to autologous ATMPs.




























To provide the best experiences, we use technologies such as cookies to store and/or access device information. Consenting to these technologies will allow us to process data such as browsing behavior or unique identifiers on this site. Not consenting or withdrawing consent may negatively affect certain features and functions.